PrEP’s Unexpected Side Effect: Reduced ‘HIV Anxiety’
When HIV first tore into America’s gay male community in the early 1980s, quotidian questions of sex, love, lust and trust transformed into weighty decisions with potential life-or-death consequences.
The decision to stop using condoms with a serious partner? Only as reliable an HIV-prevention method as your partner’s fidelity. A single instance of cheating? An indiscretion that carries the risk of an incurable and deadly disease. A random hookup? A nagging sense that, perhaps, this time was the time.
Todd Faircloth, 52, remembers those days well. In 1987, when gay men were still dying from AIDS in large numbers, Faircloth moved to New York City from North Carolina to start his big, gay life. He was just 17.
“I didn’t know anyone that lived past the age of 30, I didn’t anticipate anyone was going to live that long,” Faircloth, who now lives in Georgia with his husband, said. “It got to the point where people just assumed they all had a death sentence over their heads.”
Related
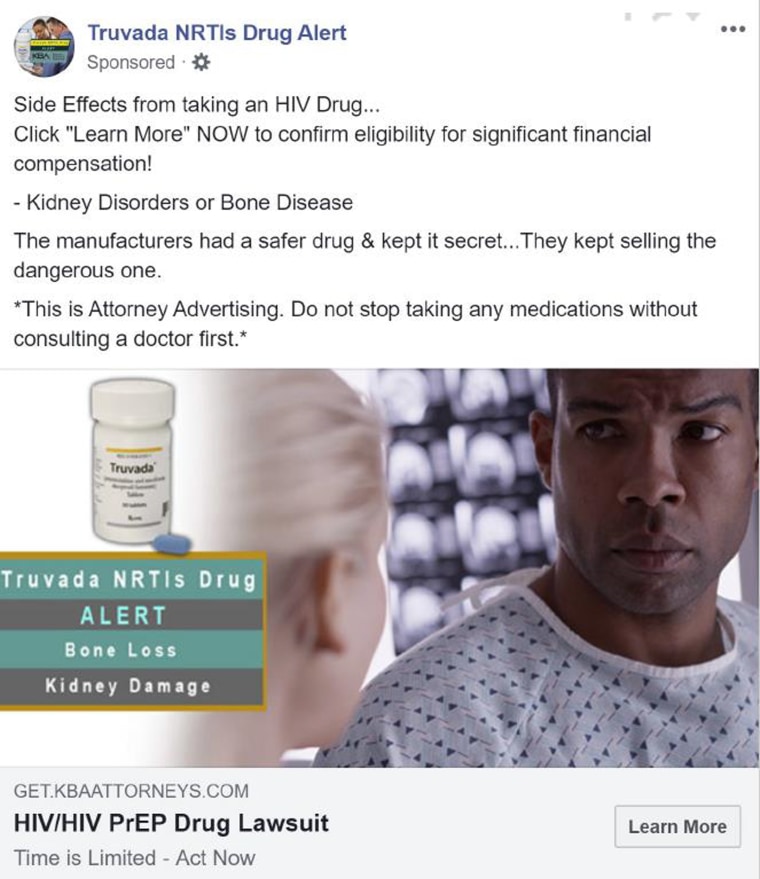
Facebook’s ‘inaccurate’ HIV PrEP ads an ‘imminent danger,’ LGBTQ groups say
He said he endured “hundreds” of AIDS funerals with a lot of dark humor, but still, “it was really scary to be out there.”
Amidst all the death, the human immunodeficiency virus caused understandable fear and anxiety among gay men, and Faircloth said this even influenced the relationships people entered into. “If you meet someone, you got with them, you were more likely to want to stay with them, not because you wanted to be with them, but because you’re scared to go back out,” he said.
Today, more than three decades after Faircloth moved to New York, HIV is controllable with medication and need not lead to death. In addition to condoms, first approved to stop HIV in 1987, people at risk of acquiring HIV today can take medications like Truvada to prevent the virus’ transmission, namely pre-exposure prophlyaxis (PrEP), and post-exposure prophylaxis (PEP), which are taken before and after sex, respectively, to prevent HIV transmission. And for those who already have the virus, treatment as prevention, or TasP, makes it impossible to transmit the virus in sex when taken regularly, according to the Centers for Disease Control and Prevention.
One unintended benefit of this new array of pharmaceutical prevention options, according to a new study, is a reduction in “HIV anxiety.” Anxiety about HIV transmission, which the study’s authors describe as a “common” experience of gay and bisexual men — especially those who, like Faircloth, lived through the darkest days of the AIDS epidemic — can compromise their “emotional well-being and create barriers to HIV testing.”by TaboolaSponsored StoriesTHE MOTLEY FOOL5 Stocks that Could Set You Up for Life2019 GIFT GUIDE27 Christmas Gifts That Are Selling Like Hot Cakes
Fifteen years ago, if the condom broke, I would be freaking out about it, and there really wouldn’t be anything you could do for three to six months except just passively hoping you don’t get HIV.
TIM PETLOCK THE OLD DAYS
“For many men, fear of HIV transmission led to anxiety about sex with other men, even in situations where transmission was impossible,” the authors note. Half of respondents worried about whether their sexual encounters were “safe,” and thought about HIV before sex, while a full quarter of study respondents, all of whom were HIV-negative, reported thinking about contracting HIV during sex. And the study found that taking PrEP “was independently associated with lower levels of HIV anxiety.”
The study suggests that this PrEP-associated reduction in HIV anxiety could “be promoted as part of demand creation initiatives to increase PrEP uptake,” a key goal of the U.S. government as it seeks to end the HIV epidemic by 2030.A sea change
Tim Petlock, 49, a gay man living near Dallas, said that so much has changed since he came out as gay in the early 1990s.
“Fifteen years ago, if the condom broke, I would be freaking out about it, and there really wouldn’t be anything you could do for three to six months except just passively hoping you don’t get HIV” Petlock told NBC News, referring to the monthslong window of early HIV tests.
“Now, you can go to treatment the day after to reduce the risk,” he said of post-exposure prophylaxis, or PEP. And today’s HIV tests can tell if a person is infected in about seven days. “You know that much sooner, whether you’ve got it or not, so it does kind of change the calculus of the whole thing.”
Today, Petlock takes PrEP and said that he worries much less about contracting HIV than he used to. Now he’s more focused on avoiding sexually transmitted bacterial infections, such as syphilis and chlamydia, which are on the rise in America.
“I know there’s some risk, but it’s likely not going to be life-altering,” he said.Ghosts of the epidemic
While PrEP has been shown to reduce HIV anxiety in some gay and bisexual men, the unease has by no means dissipated among this population.
“When you are bombarded your adult life with HIV and seeing death, I don’t care how much we advance biomedical technologies — that emotional reaction to the disease is still going to be the same,” Perry Halkitis, dean of the Rutgers University School of Public Health and author of “Out in Time,” told NBC News. “I unfortunately think that the way we deal with HIV in this country still to this day is very rooted in the responses from AIDS.”
It got to the point where people just assumed they all had a death sentence over their heads.
TODD FAIRCLOTH SURVIVING THE EPIDEMIC
One of the people for whom the memory of the epidemic’s darkest days is still fresh is Craig Lenti, a New York City-based media producer.
Lenti moved to Manhattan in 1996 at age 18 to attend college, one year after AIDS deaths peaked in the United States. He said he learned about homosexuality and AIDS at the same time, and during his late teenage years, AIDS was the top killer of young American men.
“They were always, for me, intrinsically linked,” Lenti said. “That was what I had in my head. That is what the media told me. And so from that point on, there really was no way of disconnecting those two concepts.”
Related
U.S. to give free HIV-prevention pills to uninsured — with one costly loophole
For years, Lenti avoided taking an HIV test because of his fear that it would return positive.
To this day, Lenti said, it is hard for him to take an HIV test and to trust potential partners because of his fears from the epidemic’s early days.
“It was a foregone conclusion that I would become infected,” Lenti said of his thinking at the time. Yet despite all that worrying, he remains negative.
“I think the biggest fear that I had about HIV was not the fear of being sick. It was always the fear of dying alone,” Lenti said. “It’s just very hard for me to trust people, and I think you could argue that a lot of that stems from my fear of becoming infected, even though now there’s so many different ways to combat that.”‘I don’t think it just changes overnight’
While Lenti no longer takes Truvada for PrEP because he suffered from its rare gastrointestinal side effects, he said he counsels many of his friends to take the daily medication.
“If you can take a pill that has a 99 percent effective rate, why wouldn’t you do that?” he said.
However, most at-risk Americans aren’t taking PrEP for a variety of other reasons — its high cost (a 30-day supply could cost $2,000), privacy fears and worries that the drug is dangerous (a misconception fueled in part by online ads widely criticized as deceptive). According to the CDC, PrEP is only reaching 18 percent of the 1.2 million Americans recommended to take it, and so HIV anxiety persists to this day for hundreds of thousands of gay men.
Levi, a 19-year-old college student in Ohio, is among the nearly 1 million Americans recommended to take, but not taking, PrEP. Because he’s a man who has sex with other men and is 28 or younger, he’s considered at high risk of contracting HIV, according to the CDC risk assessment worksheet for doctors (the worksheet is based on a point system and uses a variety of risk factors).
Related
12 queer artists whose work is making us pay attention
Levi is no stranger to HIV anxiety. He recalled a recent incident after he performed oral sex where he began to worry, “Do I need to get a test?”
“I spent the night basically in a hypochondriac fit running around looking if there are any sort of symptoms I should be looking for,” he said, telling an all-too-common tale. However, he needn’t have worried, as there is “little to no risk” of acquiring HIV through oral sex, according to the CDC.
Levi, who asked not to have his full name printed because he is still on his parents’ insurance and worries they would object to PrEP, said he’s just starting to think about taking the HIV prevention pill. He said the man he is dating is HIV-negative, and even though they plan to be monogamous, he doesn’t know if he can fully trust him, because the stakes are so high.
“Is there anything that I could even ask for?” Levi wondered. “Could you show me a paper or something? And could I even believe that?”
Levi’s struggle to answer this question is as old as the virus, Halkitis said.
“You have two sets of problems: a generation that has no clue and hasn’t seen death,” he said, “and a generation that is older that is completely bombarded.”
“I think there’s a whole negotiation morphing period going on right now as we get embedded in these technologies more and more,” Halkitis continued, referring to HIV prevention medications. “I don’t think it just changes overnight.”